Milk Allergies and The Paleo Diet

Dear Paleo Diet Team,
In the 1997 work entitled “Food and Low Incidence of Insulin Dependent Diabetes Mellitus (IDDM) in Iceland” (L. Thorsdottir and O. Reykdal) have suggested that the incidence of IDDM is lower in Iceland than in other human genetic related nations of Scandinavia.
Since milk proteins alleles frequencies in the Nordic cattle breeds varies and preliminary (at that time) results indicated that Beta Casein A1 was particularly low in Iceland milk, they have speculated that IDDM was caused by Beta Casein A1 and its bioactive peptide BCM-7.
My opinion is that Diabetes mellitus is not caused by one single factor alone, but perhaps there are also other factors involved.
I would like to know your opinion on that matter if possible, since cattle breeding association in Brazil, of a particular breed that produces A2 milk, is applying to official agencies to market their milk with the allegation of “non-allergenic milk.”
One of their main allegations is based on data that BCM-7 alone would be the “villain” in the milk, with which I totally disagree, since milk allergies are a multifactorial health problem.
Your attention would be greatly appreciated.
Jose Luiz M Garcia
Pedro Carrera Bastos’ Response:
Dear Mr. Garcia,
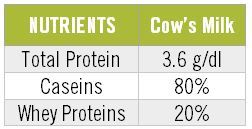
Cow’s milk (CM), as you know, has several proteins, but we can group these proteins into two major ones: whey proteins and caseins. 1 Caseins represent about 80% of the total protein content of CM, as seen in this table. 1
Caseins in CM (and also in goat’s milk) are divided into: alphaS1, alphaS2, beta and kappa.2
Following digestive proteolysis of beta-caseins in the human gut (and also after food processing, such as milk fermentation and microbial cheese ripening) there will be a release of bioactive peptides called beta-casomorphins (BCMs). 3 BCMs contain 4-11 amino acids and, more importantly, they are resistant to further proteolysis and express opioid like activity. This means they could potentially bind various opioid receptors in the human nervous system, and also in the gastrointestinal, immune and endocrine systems.3
Beta caseins (as well as the other caseins in CM) are encoded by genes found on bovine chromosome 63 and there are 12 recognized genetic variants of beta-casein: A1, A2, A3, B, C, D, E, F, H1, H2, I and G. A1 and A2 are the most common forms of beta-casein found in dairy cattle in western countries.3
Beta-caseins are proteins with 209 amino acids and the difference between A1 and A2 lies in just one amino acid at position 67: histidine in A1 and proline in A2.3, 4 Apparently, “ancestral cattle” (as well as goats, yak and most sheep) contained the A2 version of beta-casein gene and not the A1 version, which is a single nucleotide polymorphism (SNP) that appeared 5,000 to 10,000 years ago only in Bos taurus, being present today in breeds, such as Holstein, Friesian and Ayrshire.4, 5
The main premise behind the A1/A2 hypothesis is that beta-casein A1, but not A2, will originate the opioid like peptide beta-casomorphin-7 (BCM-7).3, 4 BCM-7 (which contains 7 amino acids) resists further digestion in the human gut, could be absorbed by some individuals, like babies and people with intestinal hyperpermeability, and can influence gut function without being absorbed into the bloodstream.3, 4 The proponents of this hypothesis claim that after being absorbed, BCM-7 could increase low-density lipoprotein oxidation and bind to opioid receptors in the nervous, immune and endocrine systems.3-6
This in vitro and animal data combined with epidemiological studies led to the hypothesis that beta-casein A1 is implicated in Autism and Schizophrenia,3, 4 Type 1 Diabetes,3-5 Cardiovascular Disease,3-6 Sudden Infant Death Syndrome,3 and perhaps even in Metabolic Syndrome (because it could cause insulin resistance).6
In the last years, some important scientific papers have been published criticizing the epidemiological and animal data, and particularly the lack of intervention studies supporting the above causality.5, 7, 8
Having said that, I believe the A1/A2 hypothesis should not be readily dismissed. It deserves to be better studied in animal models and, more importantly, in randomized controlled trials. Nevertheless, when it comes to Type 1 Diabetes (T1D), multiple lines of evidence strongly suggest various CM proteins, and not just beta-casein (which could yield BCM-7), are involved.
Beta-lactoglobulin (BLG)
BLG is a protein found in the whey fraction of CM (but apparently not in the whey of human’s milk) that has structural homology with the human protein glycodelin, which is responsible for the modulation of T-lymphocytes.9 This means that BLG could generate antibodies to glycodelin, and indirectly lead to autoimmunity in genetically susceptible children,9 especially if introduced early in life when there is increased intestinal permeability.9, 10
Bovine serum albumin (BSA)
This is another protein present in the whey fraction of CM. Antibodies against a specific peptide derived from BSA, called ABBOS, have been found repeatedly in the majority of patients with T1D.11-13 This is relevant because there is molecular mimicry between the peptide ABBOS and a beta-cell surface protein p69, one of the autoantigens attacked by T cells in T1D patients.11
Bovine insulin (BI)
CM, human’s milk, and presumably milk from all mammals contains insulin.10 Immunity to BI is common in children who consume cow’s milk or who have been exposed to infant formulas containing cow’s milk.10 Because BI differs from human insulin by only three amino acids, it can generate antibodies against human insulin in genetically susceptible individuals with increased intestinal permeability and other gut dysfunctions10 and/or enteral virus infections in their early years.10, 14
A recent randomized controlled trial (RCT) confirmed the role of BI in T1D.15 In this pilot trial, infants with genetic susceptibility for T1D were assigned to either a “normal” CM based formula, a whey-based hydrolyzed formula, or a whey-based formula “essentially free of bovine insulin” and it was found that the insulin-free formula reduced the cumulative incidence of autoantibodies by age 3 years.15
Interestingly, the RCT gives more support to the role of CM proteins in T1D. In this trial, 230 infants with genetic susceptibility to T1D and at least one family member with T1D received either a casein hydrolysate formula or a conventional, CM-based formula (control) whenever breast milk was not available during the first 6 to 8 months of life. The casein hydrolysate formula, as compared with the control, was associated with a decreased risk of positivity for at least one diabetes-associated autoantibody.16
In conclusion, the available evidence cannot firmly confirm or refute a causal role of BCM-7 in T1D. Nevertheless, even if a causal role is confirmed, drinking CM without beta-casein A1 could still represent a risk for people with genetically susceptibility for T1D, since there are various other potential problematic proteins in CM.
Best wishes,
Pedro Bastos, MA, MS, Ph.D. candidate in Medical Sciences at Lund University, Sweden;
References
1. Chandan RC. Milk composition, physical and processing characteristics. In Hui YH, Chandan RC, Clark S, et al. Handbook of Food Products Manufacturing – Health, Meat, Milk, Poultry, Seafood, and Vegetables. John Wiley & Sons; 2007: 347-377
2. Park YW, Haenlein GFW. Handbook of milk of non-bovine mammals. Blackwell Publishing; 2006
3. Kamiński S, Cieslińska A, Kostyra E. Polymorphism of bovine beta-casein and its potential effect on human health. J Appl Genet. 2007;48(3):189-98.
4. Woodford K. Devil in the Milk: Illness, health and politics of A1 and A2 milk. Craig Potton Publishing; 2007.
5. Merriman TR. Type 1 diabetes, the A1 milk hypothesis and vitamin D deficiency. Diab Res Clin Pract. 2008:1–8.
6. Lindeberg. Food And Western Disease: Health and Nutrition from an Evolutionary Perspective. Wiley-Blackwell; 2010.
7. Truswell AS. The A2 milk case: a critical review. Eur J Clin Nutr. 2005; 59: 623–631.
8. Clemens RA. Milk A1 and A2 peptides and diabetes. Nestle Nutr Workshop Ser Pediatr Program. 2011;67:187–195.
9. Goldfarb MF. Relation of time of introduction of cow milk protein to an infant and risk of type-1 diabetes mellitus. J Proteome Res. 2008 May;7(5):2165-7
10. Vaarala O. Is it dietary insulin? Ann N Y Acad Sci. 2006 Oct;1079:350-9.
11. Karjalainen J, et al. A bovine albumin peptide as a possible trigger of insulin-dependent diabetes mellitus. N Engl J Med. 1992 Jul 30;327(5):302-7.
12. Pérez-Bravo F, et al. Duration of breast feeding and bovine serum albumin antibody levels in type 1 diabetes: a case-control study. Pediatr Diabetes. 2003 Dec;4(4):157-61.
13. Banwell B, et al. Abnormal T-cell reactivities in childhood inflammatory demyelinating disease and type 1 diabetes. Ann Neurol. 2008 Jan;63(1):98-111.
14. Mäkelä M, et al. Enteral virus infections in early childhood and an enhanced type 1 diabetes-associated antibody response to dietary insulin. J Autoimmun. 2006 Aug;27(1):54-61.
15. Vaarala O, et al. Removal of Bovine Insulin From Cow’s Milk Formula and Early Initiation of Beta-Cell Autoimmunity in the FINDIA Pilot Study. Arch Pediatr Adolesc Med. 2012 Jul 1;166(7):608-14.
16. Knip M, et al. Dietary intervention in infancy and later signs of beta-cell autoimmunity. N Engl J Med. 2010 Nov 11;363(20):1900-8.
The Paleo Diet Team
The Paleo Diet® team consists of a group of scientists, journalists, experts, and recipe creators who stay at the forefront of nutrition science.
More About The Author




