The Science Behind Intermittent Energy Restriction

You don’t have to be on The Paleo Diet for long to start noticing extravagantly packaged, brightly colored foods that weren’t part of our Paleolithic past. But with all the time we spend explaining that hunter-gatherers didn’t eat Big Macs and jelly beans, we sometimes forget that these foods aren’t the only unnatural consequence of our modern lifestyle. Just fly across five or six time zones to see how ill equipped your body is to handle time change. As fit as our Paleolithic ancestors were, they were never able to run quite fast enough to experience jet lag.
Our bodies are, in fact, directed by circadian rhythms that dictate when we are active, when we sleep, and even when we eat. Modern conveniences such as artificial lighting and always-within-reach snacks may disturb these rhythms placing additional evolutionary stress on our bodies that ultimately affects our diet and our health. At least this is the theory presented in a recent review published in the Proceedings of the National Academy of Science by Dr. Mark Mattson et al. These researchers proposed that our health isn’t just about what we eat, but when we eat.1
Natural 24-hour rhythms permeate the entire animal and even the vegetable kingdoms.2,3 In humans, circadian oscillations have been found in over 10% of our expressed genes affecting almost all of our bodies’ metabolic, neurological and endocrine pathways.4 The suprachiasmatic nucleus (SCN) in the hypothalamus acts as our “master clock,” responding to daily light-dark cycles through photoreceptive ganglion cells in our retinas.5
Mattson et al. questioned whether the invention of artificial light and shift work perturbed this circadian clock, promoting longer daily cycles of consumption (especially nighttime meals.)
In turn, our erratic eating behaviors affect our biological clocks. Two of the main pathways in our bodies that respond to fasting and feeding—cAMP response element binding protein (CREB) for fasting and the insulin-dependent mammalian target of rapamycin (mTOR) for feeding—can directly influence circadian oscillations. In one mouse study, researchers were able to use feeding to rapidly shift liver oscillations 10 hours out of sync with the light-sensitive SCN.6
This cycle of altered eating behavior and perturbed rhythms building on one another may be a major cause of obesity and metabolic disturbances.
Which raises the question, if our natural eating pattern has been altered, is the daily western habit of three square meals truly a healthy lifestyle or a consequence of disturbed rhythms? Does it actually contribute to unnecessary metabolic stress?7-10
Recent reports of hunter-gatherer eating behavior and Dr. Cordain’s own ethnographic research paint the picture of a very different eating cycle. One of hunter-gatherers consuming a single large meal in the late afternoon or evening after spending the day hunting and gathering on little to no food.11, 12 Nor did hunter-gatherers eat consistently day-to-day. While anthropological research has debunked the notion of intermittent starvation in Paleolithic times,13 without the modern joys of a stocked fridge and preservative-packed foods, hunter-gatherers likely had frequent days of severely reduced energy intake.
So if three daily large meals puts us at evolutionary odds, how should we eat?
The health benefits of a calorie restricted (CR) diet, including a positive effect on longevity, have already been established.14-17 In their review, Mattson et al. go a step further proposing two timing-dependent variations on a CR diet that are more consistent with Paleolithic behavior and may actually enhance the health benefits.18
The first is a time-restricted feeding (TRF) diet where consumption is limited to a short period during the day—a four-to-eight-hour window—to better match life before artificial lighting. The second was intermittent energy restriction (IER) consisting of periodic days where calories are greatly reduced (i.e., just 500 calories) such as twice per week—an eating pattern that would be more consistent with a Paleolithic lifestyle. Examples of both diets are shown in the figure below:

Photo: Mattson, M.P., et al., Meal frequency and timing in health and disease. Proc Natl Acad Sci U S A, 2014. 111(47): p. 16647-53.
Benefits of Intermittent Energy Restriction
More and more studies are touting the health benefits of IER, while studies of TRF are less promising, often finding no benefits over a standard calorie restricted diet.19,20 Mattson et al. found four potential explanations for why an intermittent energy restriction diet prevents and even reverses a variety of chronic diseases including cancer, CVD, diabetes and neurodegenerative diseases.21
Better “Stress” Management
IER creates a mild stress in our bodies. But instead of being damaging, it may spark an adaptive response that ultimately enhances our defense mechanisms against more serious stress. A variety of animal studies have supported this claim showing that alternate day fasting can prevent age-related decline in the antioxidant enzymes superoxide dismutase I and catalase and can protect and even strengthen neurons against oxidative, metabolic, and proteotoxic damage.22
Improved Bioenergetic Profile
A three meal-per-day paradigm has a consequence of maintaining elevated blood sugar and insulin levels both of which have been shown to have multiple health consequences including obesity, diabetes and a variety of metabolic disorders.25-28
Due to extended periods of fasting, both IER and TRF diets improve metabolic profiles including lower blood concentrations of sugar, insulin and leptin, increased insulin sensitivity, better mobilization of fatty acids, and elevated ketones which can promote neuron health and protect against cancer.18, 29,30 The figure below shows improved insulin sensitivity and blood triglyceride concentrations on an IER diet over even a daily CR diet.
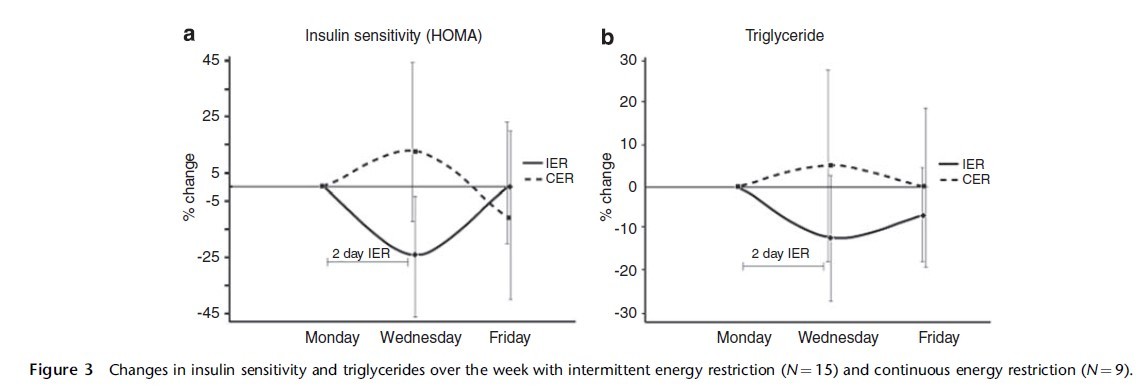
Photo: Harvie, M.N., et al., The effects of intermittent or continuous energy restriction on weight loss and metabolic disease risk markers: a randomized trial in young overweight women. Int J Obes (Lond), 2011. 35(5): p. 714-27.
Reduced Inflammation
More and more research is revealing that almost all chronic diseases are linked to inflammation both locally and throughout the body. An intermittent energy restriction diet reduces key inflammatory markers such as TNF-α and IL-6.31 However, Mattson et al. were uncertain whether the diet had a direct effect on inflammation or if it was a side effect of the weight loss typically associated with the diet.
Improved Repair and Maintenance
Damaged organelles and misfolded proteins are a natural consequence of daily living. Fortunately our cells have mechanism, such as autophagy to take up and remove damaged cellular material. Inhibition of autophagy may accelerate aging.32Eating puts the body in a protein building mode which suppresses autophagy.33 Extended periods of fasting allow our cells to switch gears and cleanse themselves.
Meal timing is a promising area of research that can bring us one step closer to eating like our Paleolithic ancestors. However, even the authors of the review pointed out that long term controlled studies are still limited and there is no consensus yet on how to best implement an IER diet.
A healthy Paleo Diet should still focus primarily on what you eat. But even if you don’t want to try a timing-dependent variation, it might be worth questioning whether 9:00 pm is the best time to eat baked salmon or if you really need that big a bowl of fruit and almonds every morning.
References
[1]Mattson, M.P., et al., Meal frequency and timing in health and disease. Proc Natl Acad Sci U S A, 2014.111(47): p. 16647-53.
[2]Reppert, S.M. and D.R. Weaver, Coordination of circadian timing in mammals. Nature, 2002. 418 (6901): p. 935-41.
[3]Goodspeed, D., et al., Postharvest circadian entrainment enhances crop pest resistance and phytochemical cycling. Curr Biol, 2013.23 (13): p. 1235-41.
[4]Panda, S., J.B. Hogenesch, and S.A. Kay, Circadian rhythms from flies to human. Nature, 2002. 417 (6886): p. 329-35.
[5]Hatori, M. and S. Panda, The emerging roles of melanopsin in behavioral adaptation to light. Trends Mol Med, 2010.16 (10): p. 435-46.
[6]Stokkan, K.A., et al., Entrainment of the circadian clock in the liver by feeding. Science, 2001. 291(5503): p. 490-3.
[7]Liu, Z., et al., PER1 phosphorylation specifies feeding rhythm in mice. Cell Rep, 2014.7(5): p. 1509-20,
[8]Hatori, M., et al., Time-restricted feeding without reducing caloric intake prevents metabolic diseases in mice fed a high-fat diet. Cell Metab, 2012. 15(6): p. 848-60.
[9]Garaulet, M., et al., Timing of food intake predicts weight loss effectiveness. Int J Obes (Lond), 2013. 37(4): p. 604-11.
[10]Vanitallie, T.B., Sleep and energy balance: Interactive homeostatic systems. Metabolism, 2006. 55 (10 Suppl 2): p. S30-5.
[11]Hill, K. and A.M. Hurtado, Aché life history : the ecology and demography of a foraging people. Foundations of human behavior. 1996, New York: Aldine de Gruyter. xix, 561 p.
[12]Berbesque, J.C., et al., Hunter-gatherers have less famine than agriculturalists. Biol Lett, 2014. 10 (1): p. 20130853.
[13]Cordain, L., J. Miller, and N. Mann, Scant evidence of periodic starvation among hunter-gatherers. Diabetologia, 1999. 42(3): p. 383-4.
[14]Fontana, L. and S. Klein, Aging, adiposity, and calorie restriction. JAMA, 2007. 297 (9): p. 986-94.
[15]Das, U.N., When less is adequate: protein and calorie restriction boosts immunity and possibly, longevity–but how and why? Nutrition, 2009. 25(9): p. 892-5.
[16]Yuan, Y., et al., Enhanced energy metabolism contributes to the extended life span of calorie-restricted Caenorhabditis elegans. J Biol Chem, 2012. 287(37): p. 31414-26.
[17]Blagosklonny, M.V., Calorie restriction: decelerating mTOR-driven aging from cells to organisms (including humans). Cell Cycle, 2010. 9 (4): p. 683-8.
[18]Harvie, M.N., et al., The effects of intermittent or continuous energy restriction on weight loss and metabolic disease risk markers: a randomized trial in young overweight women. Int J Obes (Lond), 2011. 35 (5): p. 714-27.
[19]Stote, K.S., et al., A controlled trial of reduced meal frequency without caloric restriction in healthy, normal-weight, middle-aged adults. Am J Clin Nutr, 2007.85 (4): p. 981-8.
[20]Carlson, O., et al., Impact of reduced meal frequency without caloric restriction on glucose regulation in healthy, normal-weight middle-aged men and women. Metabolism, 2007. 56(12): p. 1729-34.
[21]Longo, V.D. and M.P. Mattson, Fasting: molecular mechanisms and clinical applications. Cell Metab, 2014. 19 (2): p. 181-92.
[22]Pieri, C., et al., Food restriction in female Wistar rats: V. Lipid peroxidation and antioxidant enzymes in the liver. Arch Gerontol Geriatr, 1992. 14 (1): p. 93-9.
[23]Mattson, M.P., Energy intake and exercise as determinants of brain health and vulnerability to injury and disease. Cell Metab, 2012. 16 (6): p. 706-22.
[24]Marosi, K. and M.P. Mattson, BDNF mediates adaptive brain and body responses to energetic challenges. Trends Endocrinol Metab, 2014. 25 (2): p. 89-98.
[25]Dandona, P., A. Aljada, and A. Bandyopadhyay, Inflammation: the link between insulin resistance, obesity and diabetes. Trends Immunol, 2004. 25 (1): p. 4-7.
[26]Mehran, A.E., et al., Hyperinsulinemia drives diet-induced obesity independently of brain insulin production. Cell Metab, 2012.16 (6): p. 723-37.
[27]Brand-Miller, J.C., et al., Glycemic index and obesity. Am J Clin Nutr, 2002. 76(1): p. 281S-5S.
[28]Wang, J., et al., Overfeeding rapidly induces leptin and insulin resistance. Diabetes, 2001. 50 (12): p. 2786-91.
[29]Johnson, J.B., et al., Alternate day calorie restriction improves clinical findings and reduces markers of oxidative stress and inflammation in overweight adults with moderate asthma. Free Radic Biol Med, 2007. 42 (5): p. 665-74.
[30]Lee, C., et al., Fasting cycles retard growth of tumors and sensitize a range of cancer cell types to chemotherapy. Sci Transl Med, 2012. 4 (124): p. 124ra27.
[31]Kroeger, C.M., et al., Improvement in coronary heart disease risk factors during an intermittent fasting/calorie restriction regimen: Relationship to adipokine modulations. Nutr Metab (Lond), 2012. 9 (1): p. 98.
[32]Rubinsztein, D.C., G. Marino, and G. Kroemer, Autophagy and aging. Cell, 2011. 146 (5): p. 682-95.
[33]Speakman, J.R. and S.E. Mitchell, Caloric restriction. Mol Aspects Med, 2011. 32 (3): p. 159-221.
Trevor Connor, M.S.
Dr. Loren Cordain’s final graduate student, Trevor Connor, M.S., brings more than a decade of nutrition and physiology expertise to spearhead the new Paleo Diet team.
More About The Author